 Pure gold precipitate produced by the aqua regia chemical refining processĪqua regia dissolves gold, although neither constituent acid will do so alone. If there is contamination by dissolved metals, the neutralized solution should be collected for disposal. While local regulations may vary, aqua regia may be disposed of by careful neutralization, before being poured down the sink. īecause its components react quickly, resulting in its decomposition, aqua regia quickly loses its effectiveness (yet remains a strong acid), so its components are usually only mixed immediately before use. While chromic acid baths are discouraged because of the high toxicity of chromium and the potential for explosions, aqua regia is itself very corrosive and has been implicated in several explosions due to mishandling. This method is preferred among most over the more traditional chromic acid bath for cleaning NMR tubes, because no traces of paramagnetic chromium can remain to spoil spectra. It is also used in some laboratories to clean glassware of organic compounds and metal particles. Because nitric oxide readily reacts with atmospheric oxygen, the gases produced also contain nitrogen dioxide, NO 2 (red fume):Ģ NO O 2 → 2 NO 2 Applications Īqua regia is primarily used to produce chloroauric acid, the electrolyte in the Wohlwill process for refining the highest purity (99.999%) gold.Īqua regia is also used in etching and in specific analytic procedures. Therefore, in addition to nitrosyl chloride and chlorine, the fumes over aqua regia also contain nitric oxide (NO). This dissociation is equilibrium-limited. Nitrosyl chloride (NOCl) can further decompose into nitric oxide (NO) and elemental chlorine ( Cl 2): As the volatile products escape from solution, aqua regia loses its potency. These reactions result in the volatile products nitrosyl chloride and chlorine gas:Īs evidenced by the fuming nature and characteristic yellow color of aqua regia. Upon mixing of concentrated hydrochloric acid and concentrated nitric acid, chemical reactions occur. It was named by alchemists because it can dissolve the noble metals gold and platinum, though not all metals. Freshly prepared aqua regia is colorless, but it turns yellow, orange or red within seconds from the formation of nitrosyl chloride and nitrogen dioxide. Here, fresh aqua regia has been added to these NMR tubes to remove all traces of organic material.Īqua regia ( / ˈ r eɪ ɡ i ə, ˈ r iː dʒ i ə/ from Latin, literally "regal water" or "royal water") is a mixture of nitric acid and hydrochloric acid, optimally in a molar ratio of 1:3. So we can add/subtract in chemical equations, but its can't just simply make mathematical sense, it also has to make sense in a chemistry context.Freshly prepared aqua regia is colorless, but it turns orange within seconds. So it's not much that we're adding chemicals rather we're discovering the actual reaction occurring. However if it's an aqueous solution, these added chemicals are technically always present in the reaction solution and what we're actually doing is recognizing that some of them are actually part of the redox reaction. Now in this case where we're adding chemicals to the equation it may just seem like we're adding chemicals out of convenience to make the math work out. 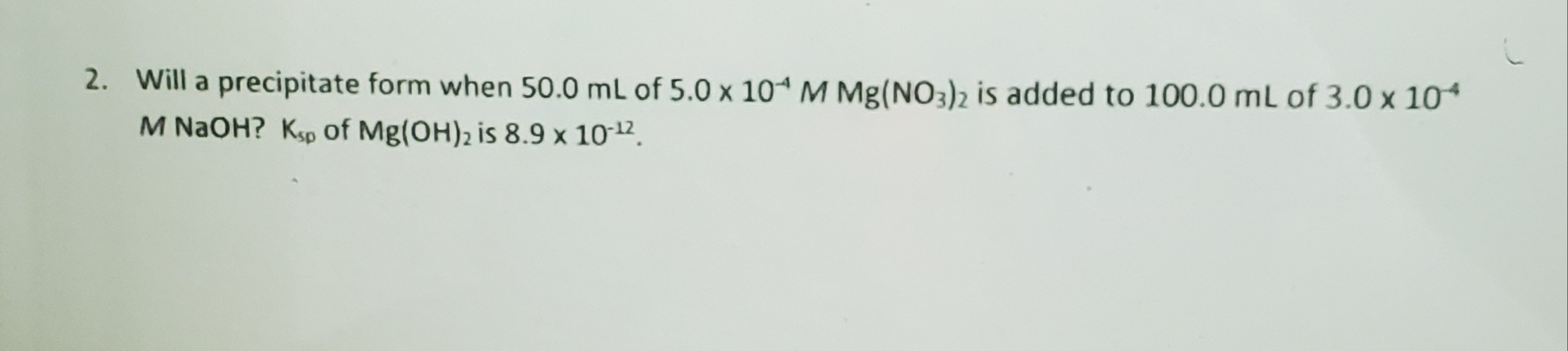
In redox reaction it's common to add water, H , and OH- ion to the equations when balancing them. So since they're not participating in the reaction, subtract them is allowed because it doesn't affect the reaction if they're absent from the equation. So these are ions which are present in the reaction solution, but don't really participate in the actual reaction (they don't change as a product compared to when they were a reactant). 
For ionic equations like these it's possible for us to eliminate, essentially subtract out, spectator ions from an equation. Mathematically it's completely acceptable to do so, however we have to consider the actual chemical makeup of our reaction if we do so. 
This reaction is represented by the molecular equation below. If we could zoom in on the contents of the reaction beaker, though, we wouldn't find actual molecules of AgNO 3 \text_4(aq) Na 2 SO 4 ( a q ) start text, N, a, end text, start subscript, 2, end subscript, start text, S, O, end text, start subscript, 4, end subscript, left parenthesis, a, q, right parenthesis. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
